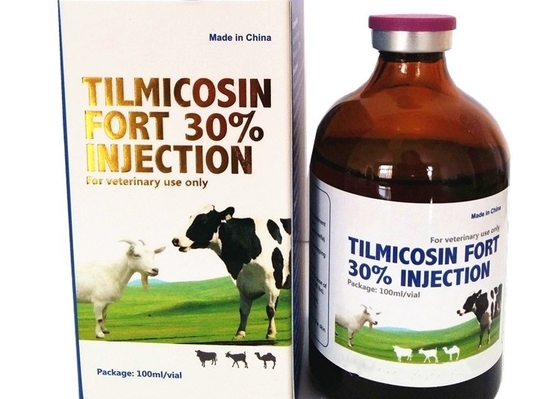
The veterinary drug Timicoxacin injection is 30% used to treat pneumonia in domestic animals
Description of Tilmicosin injection 30% :
Tilmicosin is a broad-spectrum semi-synthetic bactericidal macrolide antibiotic synthesized from tylosin. It has an antibacterial spectrum that is predominantly effective against Mycoplasma, Pasteurella and Haemophilus spp. and various Gram-positive organisms such as Corynebacterium spp. It is believed to affect bacterial protein synthesis through binding to 50S ribosomal subunits. Cross-resistance between tilmicosin and other macrolide antibiotics has been observed. Following oral administration, tilmicosin is excreted mainly via the bile into the faeces, with a small proportion being excreted via the urine.
Indication of Tilmicosin injection 30% :
For first-line control and treatment of Bovine Respiratory Disease (BRD). Also used for the treatment of respiratory bacterial diseases in large ruminants, caused by Pasteurella miltocida, Mannheimia (Pasteurella) haemolytica, infection processes associated with Haemophilus somnus, Histophilus spp. etc.
Contra indications of Tilmicosin injection 30% :
Hypersensitivity or resistance to tilmicosin.
Concurrent administration of other macrolides or lincosamides.
Administration to animals with an active microbial digestion or to equine or caprine species.
Parenteral administration, especially in porcine species.
Administration to poultry producing eggs for human consumption or to animals intended for breeding purposes.
During pregnancy and lactation, use only after a risk/benefit assessment by a veterinarian.
Side effects of Tilmicosin injection 30% :
Occasionally, a transient reduction in water or (artificial) milk intake has been observed upon treatment with tilmicosin.
| Product name |
Tilmicosin Injection 30% |
| Capacity |
10ml, 50ml, 100ml |
| Composition |
Each ml contains
Tilmicosin…………300mg |
| Indications |
Mainly used for the prevention and treatment of livestock pneumonia (caused by Actinobacillus pleuropneumoniae, Pasteurella, Mycoplasma, etc.), avian mycoplasmosis and mastitis of lactating animals. |
| Dosage and Administration |
For subcutaneous administration.
One dose, cattle: 3ml per 100kg body weight. |
| Contraindication |
Lactating cows and beef calves are prohibited. |
| Withdrawal Time |
Cattle: 35 days |
| Storage |
Store below 30℃, away from sunlight. |
| Expired Time |
2 years |
| Samples |
Available |


FAQ
1. who are we?
We are based in Henan, China, start from 2007,sell to Southeast Asia(40.00%),South Asia(10.00%),Eastern Asia(10.00%),Mid East(10.00%),Domestic Market(5.00%),South America(5.00%),Southern Europe(5.00%),Africa(5.00%),Western Europe(5.00%),Eastern Europe(5.00%). There are total about 101-200 people in our office.
2. how can we guarantee quality?
Always a pre-production sample before mass production;
Always final Inspection before shipment;
3.what can you buy from us?
Veterinary poultry medicine,antibiotic injection,animal feed additives,Veterinary Injectable Drugs,oral solution medicine
4. why should you buy from us not from other suppliers?
we possess production lines for Vaccine, Animal medicines, injection, oral liquid, Powder medicine WSP, Disinfectant, Feed additives, etc. And successfully passed the National Ministry of Agriculture in the year 2006,2011 & 2016 for GMP certification.
5. what services can we provide?
Accepted Delivery Terms: FOB,CFR,CIF;
Accepted Payment Currency:USD,EUR;
Accepted Payment Type: T/T,L/C,Western Union;
Language Spoken:English,Chinese

 Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!  Your message must be between 20-3,000 characters!
Your message must be between 20-3,000 characters! Please check your E-mail!
Please check your E-mail!